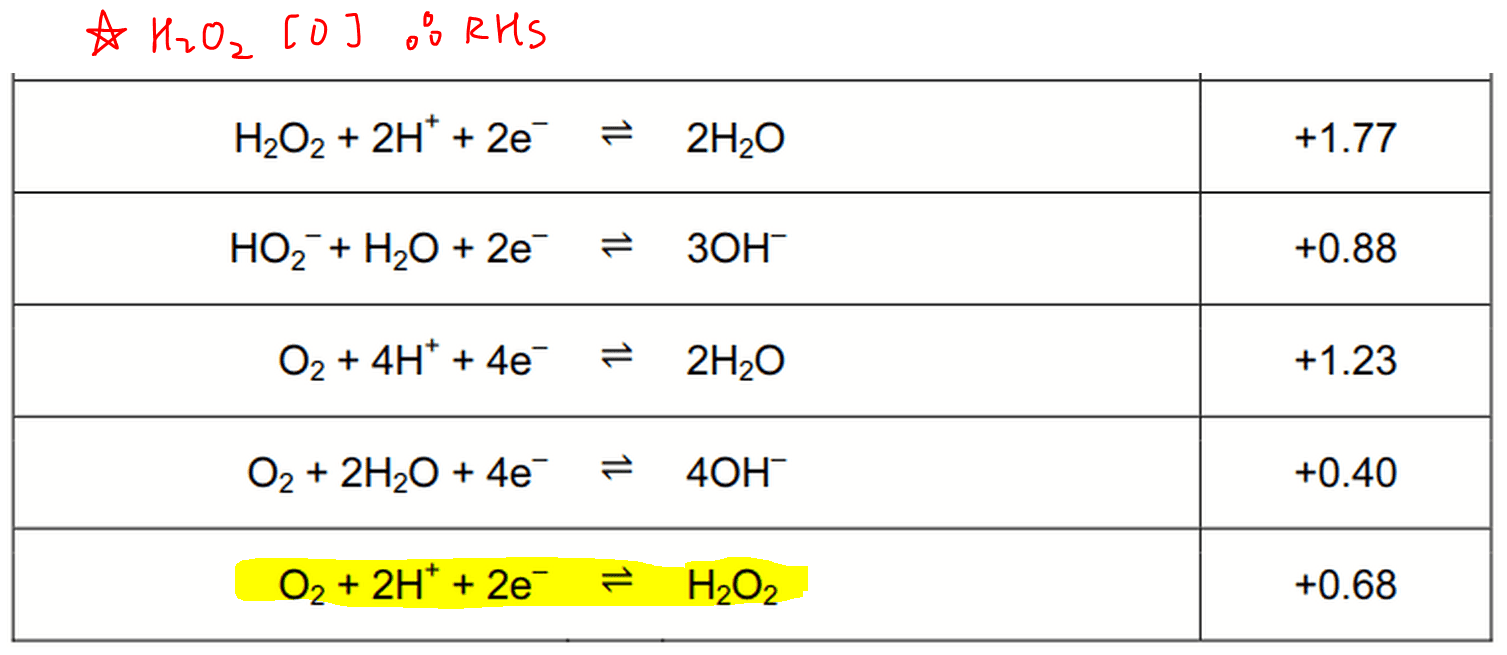
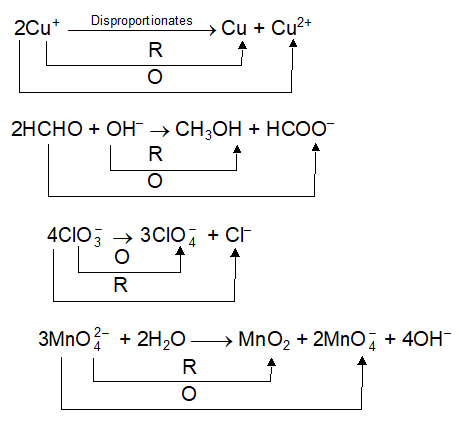
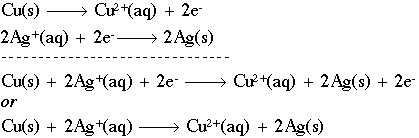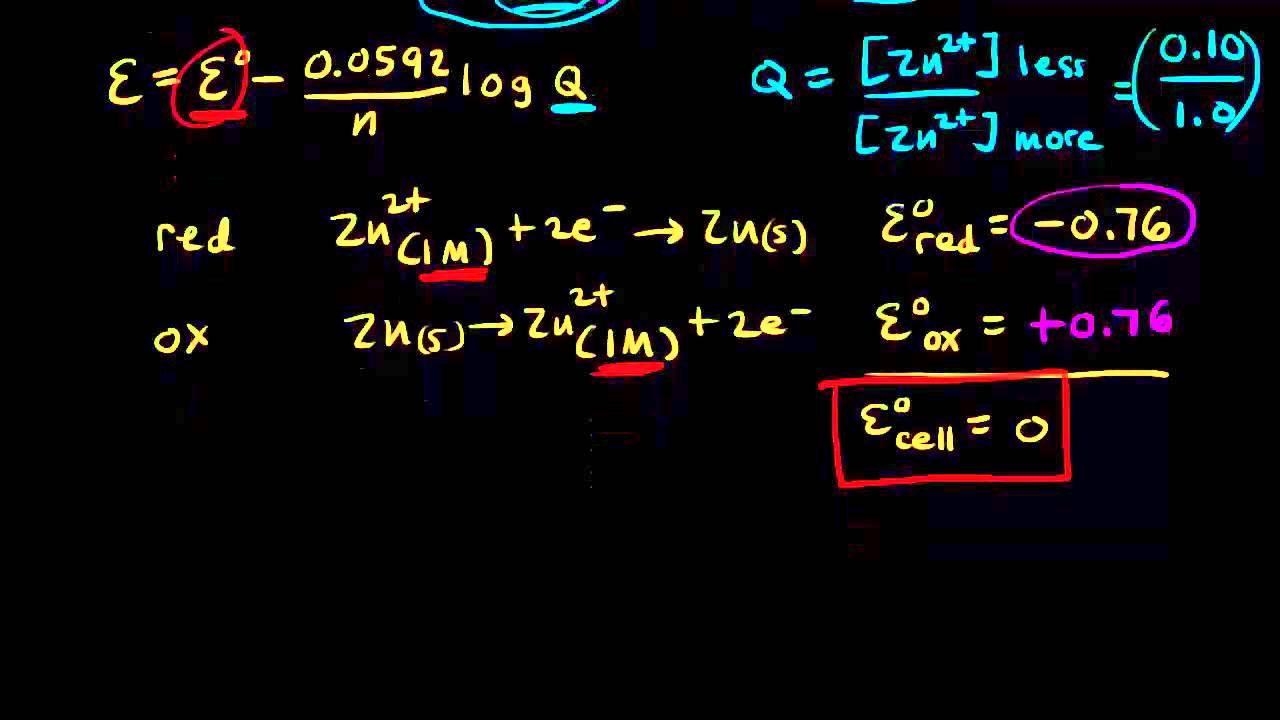Using the Nernst equation | Redox reactions and electrochemistry | Chemistry | Khan Academy - YouTube
Calculate the equilibrium constant for the redox reaction at 25°C. Sr(s) + Mg^(2+) → Sr^(2+)(aq) + Mg(s), - Sarthaks eConnect | Largest Online Education Community

equilibrium - Calculate the cathode electrode potential in this redox reaction - Chemistry Stack Exchange