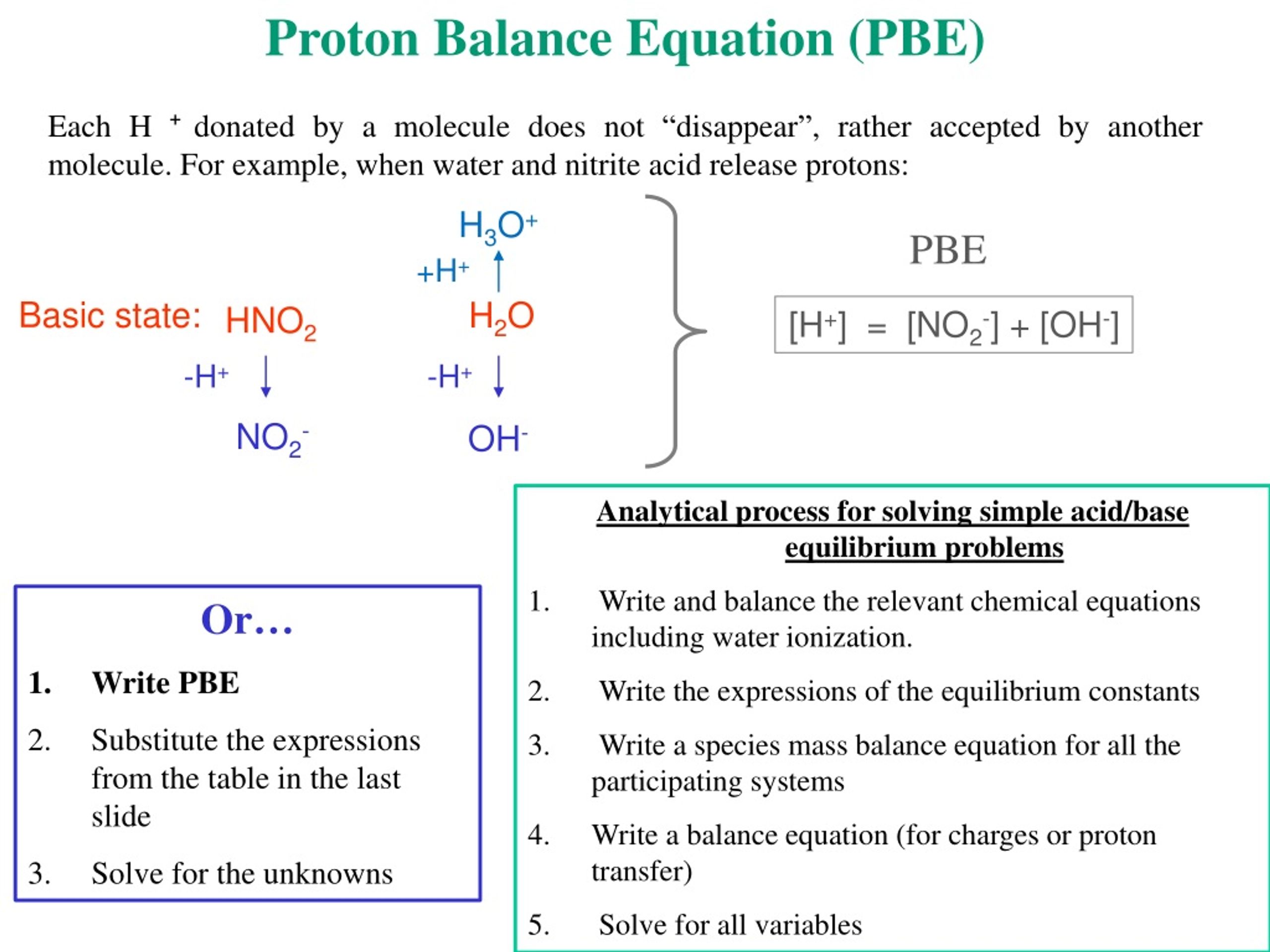
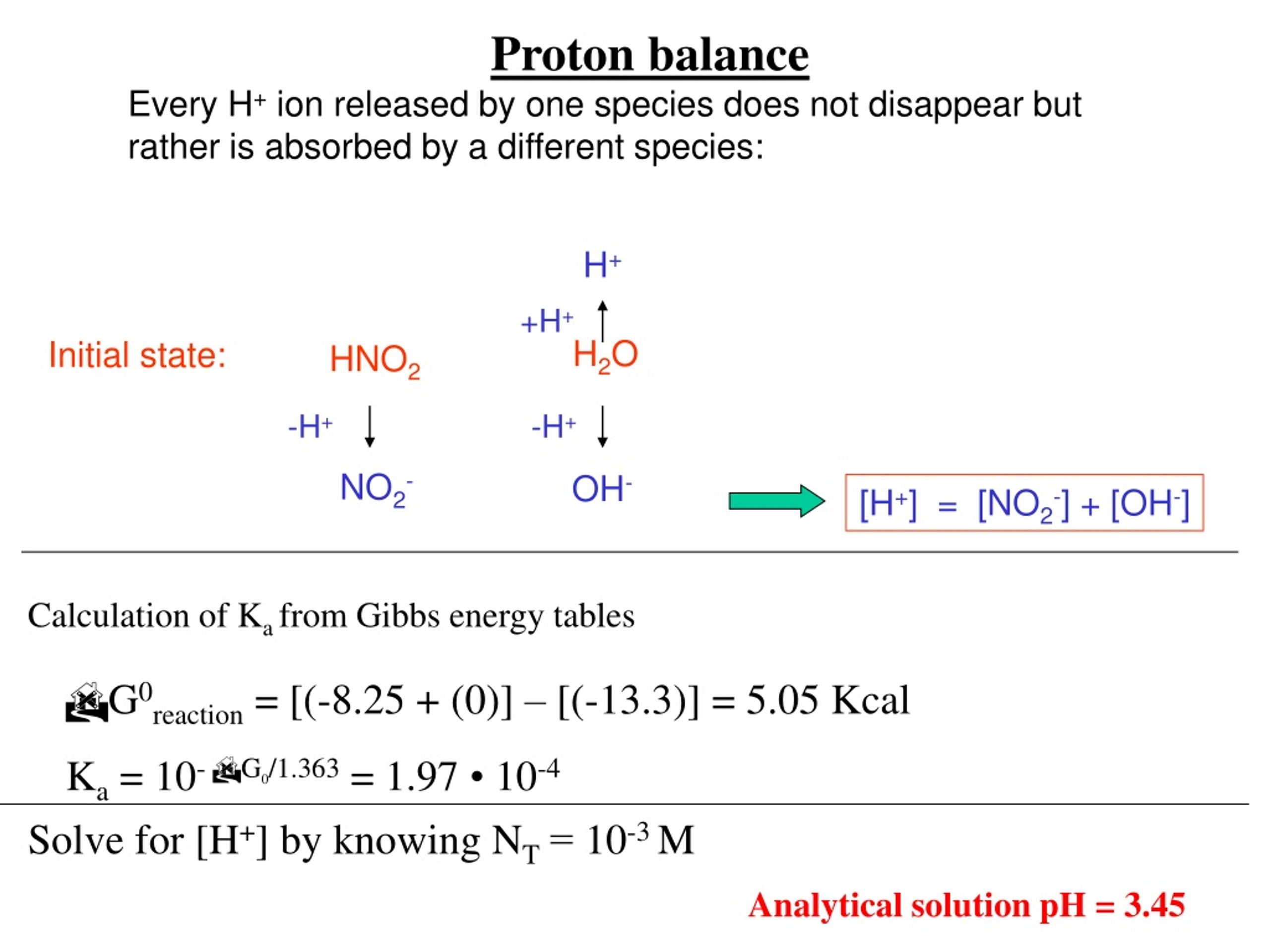
Tautomeric Equilibrium Revisited: proton-tautomerism in solvent and the fundamentals of molecular stability prediction. | Biological and Medicinal Chemistry | ChemRxiv | Cambridge Open Engage

Chapter 4(1) Acid – Base Disorders §1. Acid- Base balance 1.Concept of acid and base Acid: Acid is a proton donor. (HCl, NH 4 + ) HCl → H + + Cl - NH ppt download

SOLVED: Part III (18 points) Equations (6 points) Complete and balance the following proton transfer reactions (acid-base reactions): NOTE: To be correct, charges on ions MUST BE included. HBr (aq) + H2O (
SOLVED: Write down the mass balance, charge balance, and proton balance equations for the following solutions: NaHCO3, K2PO4, NH4HCO3, NaHCO2, 0.1 M, and H2CO3, 0.1 M.
SOLVED: 2. Write the proton balance equations when the following salts are added to water: (a) NH4Acetate (b) NaHSO4 (c) Na2SO4 (d) Na3PO4 (e) NH4HCO3

SOLVED: Write down the mass balance, charge balance, and proton balance equations for the following solutions: NaHCO3, K2PO4, NH4HCO3, NaHCO2, 0.1 M, and H2CO3, 0.1 M.

SOLVED: Write down the mass balance, charge balance, and proton balance equations for the following solutions: NaHCO3, K2PO4, NH4HCO3, NaHCO2, 0.1 M, and H2CO3, 0.1 M.